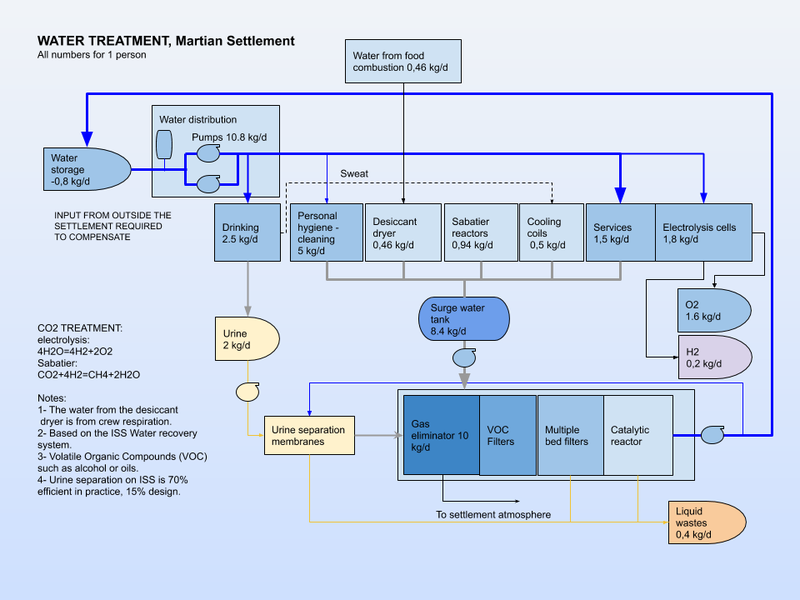
Potable water treatment
Potable water treatment facilities are an essential requirement for a Martian settlement. A number of the following processes may be required, depending on the source of the water. Some industrial processes will produce clean water than can be used in a potable water system. Waste water treatment facilities are very similar to water treatment facilities, but will treat more heavily contaminated water. In a closed loop system, the cleaned waste water is returned to the potable water treatment system for re-use. The water distribution system links the two ends of the water infrastructure.
Contents
Martian water contaminants
Salts, sand and hydrated minerals. Minerals (Ca,Mg,Fe) Primary contaminants from the martian environment and secondary contaminants from human use.
Water hardness. perchlorate.
Water treatment processes
- Reverse osmosis (perchlorates,Ca,Mg)
- Ion exchange resins (perchlorates, calcium, magnesium)
- Sand filtration (suspended solids)
- Chlorination (biological contamination, pathogens)
- Ozonation (biological contamination, pathogens)
- Activated charcoal (organics, Volatile Organic Compounds)
- PH balancing
- Electroconductivity adjustment
- Flocculation (suspended particles)
- Desalination (salts, Ca,Mg)
- Evaporation
- Organic solids removal
Water treatment plant
The first water treatment plants will be packaged units provided from Earth. These types of units are already produced on Earth for buildings and communities isolated from the larger centralized water treatment plants available for towns and cities.
Desalination
Reverse osmosis
The energy requirements of desalination for water treatment is approximately 3-3.5 kWh (electrical) per cubic meter of water, given a saltwater input equal to Earth sea water and using reverse osmosis. (2)
Evaporation
With the ~500W/m^2 sunlight intensity on Mars, an efficiency of 20-25% and a 20-25% capacity factor, a square meter of ground can produce 0.5kWh per day or sol. So each day, a solar array can desalinate enough seawater-like salty water on Mars to equal its own area up to 15-33cm deep (about 1 foot), or 150 to 300 l/m2 per day.
Production rates
New water would add itself to the recycled water in the life support systems, and compensate for losses due to water treatment required to remove dissolved salts and other contaminants.
Plants for food production and life support require large amounts of water to function correctly. In greenhouses, water irrigation requirements have a typical value of 10 to 16 l/m2 per day. So even if an open system configuration was adopted, a desalination array would be able to compensate all the required water with a production area one tenth of the farm area. If we plan for about 2% replacement of the water per cycle, then an evaporation based desalination plant would need about 500 times less area that the food production area.
TSSE
Temperature Swing Solvent Extraction(TSSE)[1]is a process for removing impurities from water using a solvent at various temperatures. It works on the basis that the amount of water that a solvent can hold changes with temperature, so by "swing"ing the temperature of the solvent, it can extract water from brine and deposit it elsewhere. This process is useful because it requires only low grade waste heat to run and easily produced industrial solvents, rather than needing high pressure pumps and custom membranes in a reverse osmosis system. The primary drawback is that without a final reverse osmosis step, the water may contain traces of solvent. A two phase system utilizing Diisopropylamine (DIPA) and diethyl ether (Eth) was suggested to resolve this issue, as the second Eth washing can remove the DIPA while being easy to remove due to the high vapor pressure and immiscibility of Eth.
References
(2) https://www.desware.net/Energy-Requirements-Desalination-Processes.aspx