Difference between revisions of "In-situ resource utilization"
| (23 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
<Imagemap> | <Imagemap> | ||
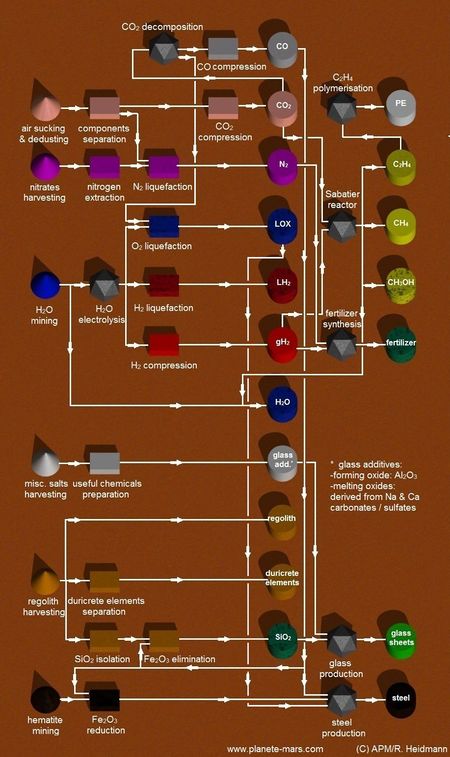
| − | File:MH ISRU.jpg|thumb|757x757px|In Situ resources Utilisation. R. Heidmann | + | File:MH ISRU.jpg|thumb|757x757px|In Situ resources Utilisation. Copyright R. Heidmann |
| + | rect 60 170 250 260 [[Dust collector]] | ||
rect 400 70 500 140 [[Compression]] | rect 400 70 500 140 [[Compression]] | ||
rect 500 70 600 140 [[Carbon monoxide]] | rect 500 70 600 140 [[Carbon monoxide]] | ||
| Line 13: | Line 14: | ||
rect 600 410 700 470 [[Sabatier process]] | rect 600 410 700 470 [[Sabatier process]] | ||
rect 720 410 820 470 [[Methane]] | rect 720 410 820 470 [[Methane]] | ||
| − | rect 60 520 | + | rect 60 520 130 590 [[water]] |
| − | rect 160 | + | rect 160 520 250 590 [[Electrolysis]] |
| − | rect 720 | + | rect 500 520 600 590 [[Hydrogen]] |
| + | rect 60 850 120 950 [[Mining]] | ||
| + | rect 60 1100 120 1200 [[Mining]] | ||
| + | rect 720 630 820 710 [[Fertilizer]] | ||
| + | rect 720 1200 820 1280 [[Glass]] | ||
| + | rect 60 1300 120 1380 [[Iron ore]] | ||
</Imagemap> | </Imagemap> | ||
Revision as of 12:42, 19 July 2019
The use of local resources is called in-situ resource utilization or ISRU. This concept is critical to the survival of an autonomous or semi-autonomous settlement.
Ressources
Atmosphere
Main article: Atmospheric processing
Many of the elements and molecules in the atmosphere can be utilized. Condensation, followed by distillation, are often used to extract resources. The atmosphere is first cooled to a liquid or solid state. This is distilled at precise temperatures in order to separate the elements and molecules.
Carbon dioxide (CO2)
Carbon dioxide composes 96% of the martian atmosphere
Carbon dioxide is the main source of carbon, used for fuel production (CH4) and an essential element for life.
Nitrogen (N2)
Nitrogen composes 2% of martian atmosphere.
Nitrogen is used by plants and is part of a breathable atmosphere. Its concentration on Earth is 78% of the atmosphere.
Argon (Ar)
2% of martian atmosphere
Argon is an inert gas, useful in some industrial processes as an inert atmosphere and may be used as propellant in Electric Propulsion of spaceships.
Water (H2O)
Water is the main source of hydrogen, used for fuel production (CH4) and for the synthesis of hydrocarbons, the building blocks for life.
Lithosphere (surface)
Main article: Mining
Minerals in the crust of Mars must be mined and processed to be useful. The upper layer of Mars surface is called the Regolith. It is a mixtures of materials of various interest.
Water
Water can be gathered in a variety of ways. It is available in the form of water ice or as hydrated minerals.
Silicates
Silicates (SiO2) are useful for the production of glass and building materials. It is one of the main components of the martian planetary crust.
Iron ore
Iron ore ( Hematite:Fe2O3) or (Magnetite: Fe3O4) is a source of iron and steel, as well as oxygen or CO2, depending on the process used.
Alumina
Alumina (Al2O3) is the source of aluminium. Processing also produces CO2 or water depending on the process used.
Carbonates
Calcium carbonate (CaCO3) is used for concrete production. Carbonates are also a potential source of carbon for carbohydrates.
Carbonates are available on Mars.[1]
Sulfates
Nitrates
Nitrates are sources of nitrogen for plants and industrial processes, ammonia and explosives. Nitrates were discovered on Mars by the Curiosity rover in 2015.
Salts
(Mg,Na)SO4, NaCl, and (Mg,Ca)CO3. Magnesium, Calcium, Sodium, lithium, Chlorine. Practically all minerals and elements can be found in the form of salts. Sodium chloride (NaCl) is the most common salt, and is essential for life.
Chlorides are likely to be abundant on MArs.[2]
Thorium
JPL has identified Thorium (Th) deposits on Mars, this is the preferred fuel in a number of Molten Salt Reactor designs. [3]
Energy
Energy is required to carry out ISRU. There are two known sources of energy on Mars, the sun and nuclear fission. Energy may be stored in a variety of ways for when the sources are not available.
Solar energy
Nuclear energy
Energy storage
Energy distribution
Embodied energy
Embodied energy is the sum of all the energy required to produce any goods or services, considered as if that energy was incorporated or 'embodied' in the product itself.[4] Embodied energy is a useful concept for the analysis of the production of martian materials, since all materials on Mars must be produced from either nuclear or solar energy.
Once the cost of energy on Mars is determined, the concept of embodied energy can be used to evaluate the cost of materials, and compared to the cost of transportation from Earth.
Processes
Compression
Mechanical compression of gases increases their density
Thermal processes
Heating and cooling are important processes that can be used to accomplish phase changes in various substances.
Crushing, milling
These are mechanical processes that break minerals down to individual crystals for separation and materials handling. Complex minerals such as basalts, granites or ores can be broken down for separation
Separation
Mechanical, centrifugal, Flottation,Distillation, Condensation
Chemical reactions
Synthesis
Hydrocarbon synthesis
- Main article: Hydrocarbon synthesis
Hydrocarbons can be manufactured by combining hydrogen and carbon through a variety of reactions.
Silicone Synthesis
- Main article: Silicone synthesis
Decomposition
Electrolysis
Deoxidation (usually, but not exclusively) of a compound into individual elements
single and double replacements.
Utilization
| This section of the article is incomplete or needs more detail. You can help Marspedia by expanding or correcting it. |
Water
Water is essential for life. It is also a common process reagent, an excellent coolant for industrial processes and a source of hydrogen and oxygen using electrolysis.
On Mars it can also be used as a construction material or as radiation shielding. It can be condensed out of the atmosphere or extracted from the regolith.
Breathable Atmosphere
A breathable atmosphere is a basic requirement for life. It is also needed for heat transfer from people, plants and animals. Is is obtained from compression of the martian atmosphere, separation of excess CO2 and addition of oxygen to reach the desired proportions, that depend on the chosen atmospheric pressure in the habitats.
Habitats
Habitats, including living and production areas, are assembled from manufactured products or possible naturally occurring areas sur as lava tubes, to create living areas for the colonists, plants and animals.
Food production
Agriculture
Plants are natural factories, capable of utilizing the atmosphere and regolith to grow and reproduce.
Manufactured Products
Propellant
Propellant is one of the main ISRU products. It is required to make transportation less prohibitively expensive.
Cements, concretes and compressed regolith
Iron and steel
Iron and steel
Aluminium
Glass
Glass is one of the most common building materials on Earth and should be common on Mars as well, since it has unique properties of low cost and transparency. Silica, the main component of glass, is also the most common material in the martian crust.
Ceramics
Ammonia fertilizer
Hydrocarbons and plastics
See Also
References
- ↑ Wikipedia Carbonates on Mars[1]
- ↑ Wikipedia- Chlorides on Mars[2]
- ↑ Map of Martian Thorium at Mid-Latitudes, JPL Map of Martian Thorium at Mid-Latitudes , https://www.jpl.nasa.gov/spaceimages/details.php?id=PIA04257, March 2003.
- ↑ https://en.wikipedia.org/wiki/Embodied_energy