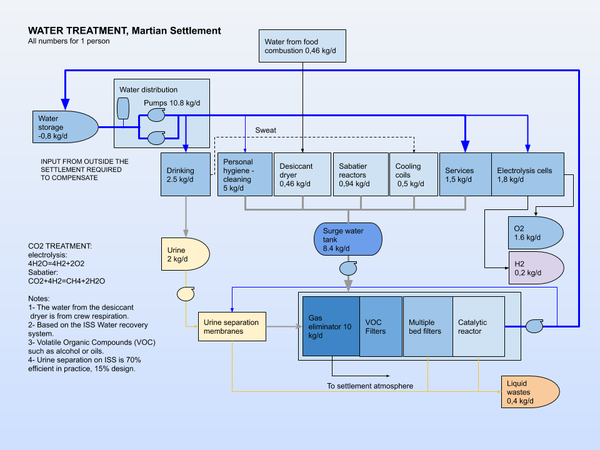
Water Infrastructure
Water, on Mars as on Earth, is essential to life and therefore to any Mars settlement. Water infrastructures, including water treatment facilities and water distribution systems will be part of any Mars settlement from day one. Indeed it will be part of the vehicles bringing the settlers to Mars.
Contents
Water extraction
Water will be available from a number of sources on Mars. However, it is not yet clear if the raw water will contain contaminants, and what will be the nature of these.
Melting
Water will mostly be in a frozen state and will need to be heated to be melted. This required about 336 kJ per kg for the phase change from ice to water. Heating the water from Mars ambient temperature to the water treatment process temperature will also require about 4,18 kJ/kg x 70C = 292 kJ/kg for a total of 628 kJ/kg. At an energy cost of 230$/GJ, this corresponds to 0,14 $/kg.
Unless waste heat is common & easily available, (say from a nuclear reactor), it is likely that ice will be warmed with inexpensive heat. This heat might be from underground greenhouses. The habitat itself is much warmer than the ambient temperature, and waste heat from the habitat itself could warm ice. Or a facility similar to a greenhouse could warm a surface area, bringing ultra cold ice to just below freezing temperature, when it is further processed. (Smart Windows would be of great help here.)
In the book, "The Case for Mars", Zubrin suggests an inexpensive way to produce water for a young exploration base. A plastic dome is placed on some ground with permafrost below it forming a fairly tight seal (so rocks must be removed). Outside mirrors reflect sunlight into this mini-greenhouse to warm the ground further. Water sublimes from below, and condenses on a cooler at one edge of the greenhouse. After the easily extracted water is removed from an area of ground, it is moved a couple meters and the process repeats. Tho slow, this system uses solar heat to do most of the work, and the water is distilled which should remove chemical impurities.
Crushing and milling
Water at freezing temperature has a hardness of about 1.5 Mohs[1]. This puts it between talc and gypsum, so relatively soft. However, ice gets harder as it gets colder. At -70C its hardness on the Mohs is about 6[2], or just below the hardness of quartz. So drilling through cold Martian ice might be very close to drilling through granite, that has a hardness of 6 to 7. However, it seems ice has significantly less shear strength than rock[3] so drilling might not be too much work, even if the ice is still cold.
Typical grinding mill operate at about 10-20 kWh per tonne[4][5], or 36 kJ/kg. With the reduced shear strength compared to rock, then crushing it might require something like 3 kJ/kg or less. This is two orders of magnitude less than the power required to melt the ice, so it is likely ice will be crushed at the source to be made transportable, but melted at the settlement, ideally using waste heat.
Transportation
Transportation of water on Mars will probably mostly be done using heavy trucks. These use about 2,4 kJ/kg/km on Earth.[6]. For a water source 15 km away, the energy to move the ice will be similar to the energy required to extract the ice, but still less than 5% of the energy required to melt the ice. Rail might reduce the energy significantly, and the lower Martian gravity might help as well.
Recycled water will incur none of the above costs. So water treatment is interesting as an energy efficiency measure at the settlement, depending of the cost of the recycling equipment, which may be quite high if it comes from Earth.
Water treatment plants
- Extracted water may contain contaminants such as dust, salts and other materials.
- Potable water will need treatment to control biological contaminants (chlorine).
- Water may (will) be mixed with re-used water. There will be large amounts of water removed by dehumidification from greenhouses or underground grow rooms.
Water storage and distribution
Water is usually stored in tanks and distributed through pipes using pumps. Raw water is water that has been extracted, but not yet treated for use.
Water production
Although most water will be extracted from external sources, water will be produced from two sources in a settlement:
- Respiration and biological processes produce water through combustion of food in the organism.
- Propellant production produces water as part of the Sabatier process output. For every methane molecule produced, four water molecules are required, and two water molecules are returned. 4H2O+CO2=2H2O+O2+CH4.
As production increases in a growing settlement, water may become an output from the fabrication of steel if a hydrogen reduction system is used.
Water usage
- Water is used for life support and in agriculture and food production in general.
- Water is often used as a medium in cooling and heating systems. It can be reused after light treatment depending on contamination from the processes, or used in closed loops.
- Water is a source of hydrogen for a multitude of processes.
- Water is used for cleaning and dust control. Water may be used to clean equipment and remove perchlorates.
- Water is often used for fire protection is sprinkler systems.
- Sanitation systems usually use water to transport wastes. Alternatives exist.
- Water is used in a multitude of chemical reactions.
- Water is often used in concentrating metal ores before final refining.
- Water may be used on Mars to act as a thermal buffer, stabilizing the colony's temperature.
- Water may be used for recreation (e.g. swimming).
- Water is a very useful form of radiation protection. The hydrogen atoms in its structure are very good at stopping small particle radiation.
A plentiful source of water is endlessly useful for a new colony. Colonizing a world where water is hard to extract (e.g. Venus), will cause endless difficulties and added expense.
Waste water treatment
Used water goes to the waste water treatment plant. It is purified using different treatments and the minerals and contaminants removed are recycled. The water is stored, or mixed with water from the settlement systems and returned to water treatment plant for reuse.
However, depending on the cost of water treatment, it may be more cost effective to return more heavily contaminated water to the Martian environment, possible recuperating the contaminants as resources when the water evaporates in the low pressure Martian atmosphere. Or such water may be put into a plastic container and frozen, then stacked around or under the settlement to act as a Radiation shield. (Water is very good at slowing down small particles such as Cosmic radiation, and neutrons.)
References
- ↑ https://en.wikipedia.org/wiki/Mohs_scale_of_mineral_hardness
- ↑ http://academic.emporia.edu/aberjame/ice/lec02/lec2.htm
- ↑ https://www.researchgate.net/figure/Compressive-strength-and-density-of-ice-versus-a-variety-of-rock-types-data-from_fig3_272743517
- ↑ IMPROVING ENERGY EFFICIENCY VIA OPTIMIZED CHARGE MOTION AND SLURRY FLOW IN PLANT SCALE SAG MILLS
- ↑ Hard Rock Miner's handbookhttps://www.stantec.com/content/dam/stantec/files/PDFAssets/2014/Hard%20Rock%20Miner%27s%20Handbook%20Edition%205_3.pdf
- ↑ https://en.wikipedia.org/wiki/Energy_efficiency_in_transport